
Vinorelbine
Pharmaceutical drug / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Vinorelbine?
Summarize this article for a 10 year old
Vinorelbine (NVB), sold under the brand name Navelbine among others, is a chemotherapy medication used to treat a number of types of cancer.[4] This includes breast cancer and non-small cell lung cancer.[4] It is given by injection into a vein or by mouth.[4][1]
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Navelbine |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a695013 |
| Pregnancy category |
|
| Routes of administration | intravenous, by mouth[1] |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | 43 ± 14% (oral)[3] |
| Protein binding | 79 to 91% |
| Metabolism | liver (CYP3A4-mediated) |
| Elimination half-life | 27.7 to 43.6 hours |
| Excretion | Fecal (46%) and kidney (18%) |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| DrugBank |
|
| ChemSpider |
|
| UNII | |
| KEGG |
|
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
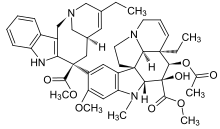
| Formula | C45H54N4O8 |
| Molar mass | 778.947 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
Common side effects include bone marrow suppression, pain at the site of injection, vomiting, feeling tired, numbness, and diarrhea.[4] Other serious side effects include shortness of breath.[4] Use during pregnancy may harm the baby.[4] Vinorelbine is in the vinca alkaloid family of medications.[1] It is believed to work by disrupting the normal function of microtubules and thereby stopping cell division.[4]
Vinorelbine was approved for medical use in the United States in 1994.[4] It is on the World Health Organization's List of Essential Medicines.[5][6]