
Uranium hexachloride
Chemical compound / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Uranium hexachloride?
Summarize this article for a 10 year old
SHOW ALL QUESTIONS
Uranium hexachloride (UCl6) is an inorganic chemical compound of uranium in the +6 oxidation state.[1][2] UCl6 is a metal halide composed of uranium and chlorine. It is a multi-luminescent dark green crystalline solid with a vapor pressure between 1-3 mmHg at 373.15 K.[3] UCl6 is stable in a vacuum, dry air, nitrogen and helium at room temperature. It is soluble in carbon tetrachloride (CCl4). Compared to the other uranium halides, little is known about UCl6.
Quick Facts Names, Identifiers ...
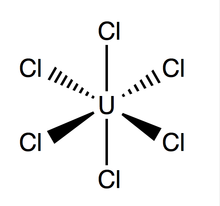 | |
| Names | |
|---|---|
| IUPAC name
Uranium(VI) chloride | |
| Other names
Uranium hexachloride Peruranic chloride | |
| Identifiers | |
3D model (JSmol) |
|
| ChemSpider | |
PubChem CID |
|
| |
| |
| Properties | |
| UCl6 | |
| Molar mass | 450.745 g/mol |
| Appearance | dark green crystalline solid |
| Density | 3.6 g/cm3 |
| Melting point | 177 °C (351 °F; 450 K) |
| Related compounds | |
Other anions |
Uranium hexafluoride |
Other cations |
Tungsten hexachloride |
Related uranium chlorides |
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Close