
Tungsten hexabromide
Chemical compound / From Wikipedia, the free encyclopedia
Tungsten hexabromide, also known as tungsten(VI) bromide, is a chemical compound of tungsten and bromine with the formula WBr6. It is an air-sensitive dark grey powder that decomposes above 200 °C to tungsten(V) bromide and bromine.[1][3]
Quick Facts Names, Identifiers ...
 | |
| Names | |
|---|---|
Other names
| |
| Identifiers | |
3D model (JSmol) |
|
| ChemSpider | |
PubChem CID |
|
| |
| |
| Properties | |
| WBr6 | |
| Molar mass | 663.264 g/mol |
| Appearance | Dark grey solid |
| Density | 5.32 g/cm3 |
| Melting point | 232 °C (450 °F; 505 K) (decomposition) |
| Hydrolysis | |
| Solubility | Soluble in ethanol, ether, carbon disulfide, and ammonia[1] |
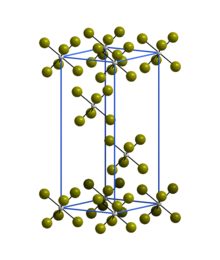
| Structure[2] | |
| Rhombohedral | |
| R3 | |
a = 6.39 Å, c = 17.53 Å | |
Lattice volume (V) |
620.8 Å3 |
Formula units (Z) |
3 |
| Related compounds | |
Other anions |
Tungsten hexafluoride Tungsten hexachloride |
Related compounds |
Tungsten(V) bromide |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Close