
Trimethylphosphine
Chemical compound / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Trimethylphosphine?
Summarize this article for a 10 year old
SHOW ALL QUESTIONS
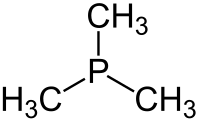
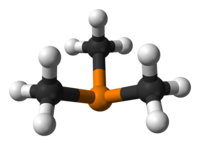
Trimethylphosphine is an organophosphorus compound with the formula P(CH3)3, commonly abbreviated as PMe3. This colorless liquid has a strongly unpleasant odor, characteristic of alkylphosphines. The compound is a common ligand in coordination chemistry.
Quick Facts Names, Identifiers ...
 | |
 | |
 | |
| Names | |
|---|---|
| Preferred IUPAC name
Trimethylphosphane | |
| Systematic IUPAC name | |
| Identifiers | |
| |
3D model (JSmol) |
|
| 969138 | |
| ChEBI | |
| ChemSpider |
|
| ECHA InfoCard | 100.008.932 |
| EC Number |
|
| MeSH | trimethyl+phosphine |
PubChem CID |
|
| UNII | |
| UN number | 1993 |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C3H9P | |
| Molar mass | 76.079 g·mol−1 |
| Appearance | Colorless liquid |
| Density | 735 mg cm−3 |
| Melting point | −86 °C (−123 °F; 187 K) |
| Boiling point | 38 to 39 °C (100 to 102 °F; 311 to 312 K) |
| Vapor pressure | 49.9 kPa (at 20 °C) |
| Structure | |
| Trigonal pyramidal | |
| 1.19 Debye | |
| Hazards | |
| GHS labelling: | |
  | |
| Danger | |
| H225, H315, H319, H335 | |
| P210, P261, P305+P351+P338 | |
| Flash point | −19 °C (−2 °F; 254 K) |
| Related compounds | |
Related compounds |
PEt3 NMe3 PH3 PPh3 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Close