
Tartrate
Chemical compound / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Tartrate?
Summarize this article for a 10 year old
SHOW ALL QUESTIONS
A tartrate is a salt or ester of the organic compound tartaric acid, a dicarboxylic acid. The formula of the tartrate dianion is O−OC-CH(OH)-CH(OH)-COO− or C4H4O62−.[1]
Quick Facts Identifiers, Properties ...
 | |
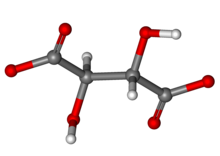 | |
| Identifiers | |
|---|---|
3D model (JSmol) |
|
| ChEBI | |
| ChemSpider |
|
PubChem CID |
|
| UNII | |
| |
| |
| Properties | |
| C4H4O62− | |
| Molar mass | 148.07 g/mol |
| Conjugate acid | Bitartrate |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Close
The main forms of tartrates used commercially are pure crystalline tartaric acid used as an acidulant in non-alcoholic drinks and foods, cream of tartar used in baking, and Rochelle salt, commonly used in electroplating solutions.