Sodium methylsulfinylmethylide
Chemical compound / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Sodium methylsulfinylmethylide?
Summarize this article for a 10 year old
SHOW ALL QUESTIONS
Sodium methylsulfinylmethylide (also called NaDMSO or dimsyl sodium) is the sodium salt of the conjugate base of dimethyl sulfoxide. This unusual salt has some uses in organic chemistry as a base and nucleophile.
Quick Facts Names, Identifiers ...
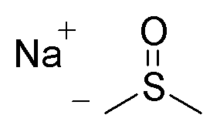 | |
| Names | |
|---|---|
| Preferred IUPAC name
Sodium (methanesulfinyl)methanide | |
| Other names
sodium dimsylate, dimsylsodium, NaDMSYL | |
| Identifiers | |
3D model (JSmol) |
|
| Abbreviations | NaDMSO |
| ChemSpider |
|
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C2H5NaOS | |
| Molar mass | 100.13 |
| Appearance | White solid, solution in DMSO is green |
| decomposes | |
| Solubility | Very soluble in DMSO and many polar organic solvents |
| Hazards | |
| Occupational safety and health (OHS/OSH): | |
Main hazards |
May form corrosive NaOH, May be explosive in certain circumstances.[1] |
| Related compounds | |
Related compounds |
Dimethyloxosulfonium methylide, dimethyl sulfoxide |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Close
Since the first publication in 1965 by Corey et al.,[2] a number of additional uses for this reagent have been identified.[3]