
Rubidium sulfide
Chemical compound / From Wikipedia, the free encyclopedia
Rubidium sulfide is an inorganic compound and a salt with the chemical formula Rb2S. It is a white solid with similar properties to other alkali metal sulfides.
Quick Facts Names, Identifiers ...
 | |
| Names | |
|---|---|
| IUPAC name
Rubidium sulfide | |
| Identifiers | |
3D model (JSmol) |
|
PubChem CID |
|
| |
| |
| Properties | |
| Rb2S | |
| Molar mass | 203.00 |
| Appearance | white crystal |
| Density | 2.912 g/cm3[1] |
| Melting point | 530 °C[2] |
| hydrolyses to rubidium bisulfide[1] | |
| Solubility in ethanol and glycerol | soluble |
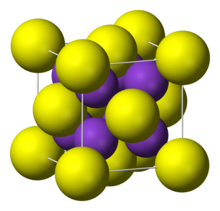
| Structure | |
| cubic:anti-fluorite | |
| Hazards | |
| Occupational safety and health (OHS/OSH): | |
Main hazards |
toxic |
| GHS labelling: | |
  | |
| Danger | |
| H314, H400 | |
| P260, P264, P273, P280, P301+P330+P331, P303+P361+P353, P304+P340, P305+P351+P338, P310, P321, P363, P391, P405, P501 | |
| Related compounds | |
Other anions |
Rubidium oxide Rubidium selenide Rubidium telluride Rubidium polonide |
Other cations |
Lithium sulfide Sodium sulfide Potassium sulfide Caesium sulfide Francium sulfide |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Close