
Ritonavir
Antiretroviral medication / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Ritonavir?
Summarize this article for a 10 year old
Ritonavir, sold under the brand name Norvir, is an antiretroviral medication used along with other medications to treat HIV/AIDS.[4][5][6] This combination treatment is known as highly active antiretroviral therapy (HAART).[6] Ritonavir is a protease inhibitor, though it now mainly serves to boost the potency of other protease inhibitors.[6][7] It may also be used in combination with other medications to treat hepatitis C and COVID-19.[8][9] It is taken by mouth.[6] Tablets of ritonavir are not bioequivalent to capsules, as the tablets may result in higher peak plasma concentrations.[6]
 | |
 | |
| Clinical data | |
|---|---|
| Pronunciation | /rɪˈtɒnəˌvɪər/ rih-TO-nə-veer |
| Trade names | Norvir |
| Other names | RTV |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a696029 |
| License data | |
| Pregnancy category |
|
| Routes of administration | By mouth |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Protein binding | 98–99% |
| Metabolism | Liver |
| Elimination half-life | 3–5 hours |
| Excretion | Mostly fecal |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| NIAID ChemDB | |
| PDB ligand | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.125.710 |
| Chemical and physical data | |
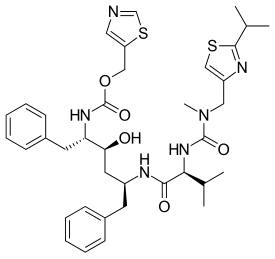
| Formula | C37H48N6O5S2 |
| Molar mass | 720.95 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Common side effects of ritonavir include nausea, vomiting, loss of appetite, diarrhea, and numbness of the hands and feet.[6] Serious side effects include liver complications, pancreatitis, allergic reactions, and arrythmias.[6] Serious interactions may occur with a number of other medications including amiodarone and simvastatin.[6] At low doses, it is considered to be acceptable for use during pregnancy.[10] Ritonavir is of the protease inhibitor class.[6] However, it is also commonly used to inhibit the enzyme that metabolizes other protease inhibitors.[11] This inhibition allows lower doses of these latter medications to be used.[11]
Ritonavir was patented in 1989 and came into medical use in 1996.[12][13] It is on the World Health Organization's List of Essential Medicines.[14][15] Ritonavir capsules were approved as a generic medication in the United States in 2020.[16]