
Risperidone
Antipsychotic medication / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Risperidone?
Summarize this article for a 10 year old
Risperidone, sold under the brand name Risperdal among others, is an atypical antipsychotic[2] used to treat schizophrenia and bipolar disorder.[2] It is taken either by mouth or by injection (subcutaneous or intramuscular).[2] The injectable versions are long-acting and last for 2–4 weeks.[9]
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Risperdal, Okedi, others[1] |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a694015 |
| License data |
|
| Pregnancy category |
|
| Routes of administration | By mouth, intramuscular, subcutaneous |
| Drug class | Atypical antipsychotic[2] |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | 70% (by mouth)[2] |
| Metabolism | Liver (CYP2D6 mediated to 9-hydroxyrisperidone)[2] |
| Elimination half-life | 20 hours (by mouth), 3–6 days (IM)[2] |
| Excretion | Urinary (70%) feces (14%)[2] |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| PubChem SID | |
| IUPHAR/BPS | |
| DrugBank |
|
| ChemSpider |
|
| UNII | |
| KEGG |
|
| ChEBI | |
| ChEMBL |
|
| PDB ligand | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.114.705 |
| Chemical and physical data | |
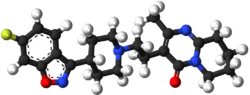
| Formula | C23H27FN4O2 |
| Molar mass | 410.493 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| (verify) | |
Common side effects include severe depression, movement problems, sleepiness, dizziness, trouble seeing, constipation, and increased weight.[2][10] Serious side effects may include the potentially permanent movement disorder tardive dyskinesia, as well as neuroleptic malignant syndrome, an increased risk of suicide, and high blood sugar levels.[2][9] In older people with psychosis as a result of dementia, it may increase the risk of death.[2] It is unknown if it is safe for use in pregnancy.[2] Its mechanism of action is not entirely clear, but is believed to be related to its action as a dopamine and serotonin antagonist.[2]
Study of risperidone began in the late 1980s and it was approved for sale in the United States in 1993.[2][11][7] It is on the World Health Organization's List of Essential Medicines.[12] It is available as a generic medication.[9] In 2021, it was the 138th most commonly prescribed medication in the United States, with more than 4 million prescriptions.[13][14]