Potassium perchlorate
Chemical compound / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Potassium perchlorate?
Summarize this article for a 10 year old
SHOW ALL QUESTIONS
Potassium perchlorate is the inorganic salt with the chemical formula KClO4. Like other perchlorates, this salt is a strong oxidizer although it usually reacts very slowly with organic substances. This, usually obtained as a colorless, crystalline solid, is a common oxidizer used in fireworks, ammunition percussion caps, explosive primers, and is used variously in propellants, flash compositions, stars, and sparklers. It has been used as a solid rocket propellant, although in that application it has mostly been replaced by the higher performance ammonium perchlorate.
Quick Facts Names, Identifiers ...
 | |||
| |||
 | |||
| Names | |||
|---|---|---|---|
| Other names
Potassium chlorate(VII); Perchloric acid, potassium salt; peroidin | |||
| Identifiers | |||
3D model (JSmol) |
|||
| ChEMBL | |||
| ChemSpider |
| ||
| ECHA InfoCard | 100.029.011 | ||
| EC Number |
| ||
PubChem CID |
|||
| RTECS number |
| ||
| UNII | |||
| UN number | 1489 | ||
CompTox Dashboard (EPA) |
|||
| |||
| |||
| Properties | |||
| KClO4 | |||
| Molar mass | 138.55 g/mol | ||
| Appearance | colourless/ white crystalline powder | ||
| Density | 2.5239 g/cm3 | ||
| Melting point | 610 °C (1,130 °F; 883 K) decomposes from 400 °C[1][2] | ||
| 0.76 g/100 mL (0 °C) 1.5 g/100 mL (25 °C)[3] 4.76 g/100 mL (40 °C) 21.08 g/100 mL (100 °C)[4] | |||
Solubility product (Ksp) |
1.05·10−2[5] | ||
| Solubility | negligible in alcohol insoluble in ether | ||
| Solubility in ethanol | 47 mg/kg (0 °C) 120 mg/kg (25 °C)[4] | ||
| Solubility in acetone | 1.6 g/kg[4] | ||
| Solubility in ethyl acetate | 15 mg/kg[4] | ||
Refractive index (nD) |
1.4724 | ||
| Structure | |||
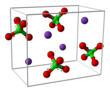
| Rhombohedral | |||
| Thermochemistry | |||
Heat capacity (C) |
111.35 J/mol·K[6] | ||
Std molar entropy (S⦵298) |
150.86 J/mol·K[6] | ||
Std enthalpy of formation (ΔfH⦵298) |
-433 kJ/mol[7] | ||
Gibbs free energy (ΔfG⦵) |
-300.4 kJ/mol[4] | ||
| Hazards | |||
| GHS labelling: | |||
  [2] [2] | |||
| Danger | |||
| H271, H302, H335[2] | |||
| P220, P280[2] | |||
| NFPA 704 (fire diamond) | |||
| Safety data sheet (SDS) | MSDS | ||
| Related compounds | |||
Other anions |
Potassium chloride Potassium chlorate Potassium periodate | ||
Other cations |
Ammonium perchlorate Sodium perchlorate | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
Close