
Polyaspartic acid
Chemical compound / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Polyaspartic acid?
Summarize this article for a 10 year old
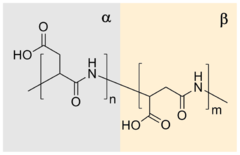
Polyaspartic acid (PASA) is a biodegradable, water-soluble condensation polymer based on the amino acid aspartic acid.[1][2] It is a biodegradable replacement for water softeners and related applications.[3] PASA can be chemically crosslinked with a wide variety of methods to yield PASA hydrogels.[4] The resulting hydrogels are pH-sensitive such that under acidic conditions, they shrink, while the swelling capacity increases under alkaline conditions.[4]
 | |
| Names | |
|---|---|
| Other names
PASP | |
| Identifiers | |
| |
| ChemSpider |
|
CompTox Dashboard (EPA) |
|
| Properties | |
| (C4H5NO3)n | |
| Molar mass | variable |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |

Sodium polyaspartate is a sodium salt of polyaspartic acid.
In nature, PASA has been found in as fragments of larger proteins with length up to 50 amino acids,[5] but as of 2004 had not been isolated as a pure homo polymeric material from any natural source.[6] The first isolation of synthetic oligomeric sodium polyaspartate, obtained by thermal polycondensation of aspartic acid, was reported by Hugo Schiff in late 19th century.[7] Later it was proposed that thermal polymerization process leads through polysuccinimide intermediate.[8][9] Polyaspartic acid is produced industrially in both the acid form and as the sodium salt.[2]