Phosphoric acid
Chemical compound (PO(OH)3) / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Phosphoric acid?
Summarize this article for a 10 year old
 | |||
| |||
| Names | |||
|---|---|---|---|
| IUPAC name
Phosphoric acid | |||
| Other names
Orthophosphoric acid | |||
| Identifiers | |||
3D model (JSmol) |
|||
| ChEBI | |||
| ChEMBL | |||
| ChemSpider |
| ||
| ECHA InfoCard | 100.028.758 | ||
| EC Number |
| ||
| E number | E338 (antioxidants, ...) | ||
| KEGG |
| ||
PubChem CID |
|||
| RTECS number |
| ||
| UNII | |||
| UN number | 1805 | ||
CompTox Dashboard (EPA) |
|||
| |||
| |||
| Properties | |||
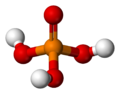
| H3PO4 | |||
| Molar mass | 97.994 g·mol−1 | ||
| Appearance | Colorless solid | ||
| Odor | Odorless | ||
| Density | 1.6845 g/cm3 (25 °C, 85%),[1] 1.834 g/cm3 (solid)[2] | ||
| Melting point | 42.35 °C (108.23 °F; 315.50 K) anhydrous[3] 29.32 °C (84.78 °F; 302.47 K) hemihydrate[4] | ||
| Boiling point | |||
| Solubility | Soluble in ethanol | ||
| log P | −2.15[9] | ||
| Vapor pressure | 0.03 mmHg (20 °C)[10] | ||
| Conjugate base | Dihydrogen phosphate | ||
| −43.8·10−6 cm3/mol[11] | |||
Refractive index (nD) |
| ||
| Viscosity | 2.4–9.4 cP (85% aq. soln.) 147 cP (100%) | ||
| Structure | |||
| Monoclinic | |||
| Tetrahedral | |||
| Thermochemistry[13] | |||
Heat capacity (C) |
145.0 J/(mol⋅K) | ||
Std molar entropy (S⦵298) |
150.8 J/(mol⋅K) | ||
Std enthalpy of formation (ΔfH⦵298) |
−1271.7 kJ/mol | ||
Gibbs free energy (ΔfG⦵) |
−1123.6 kJ/mol | ||
| Hazards | |||
| GHS labelling: | |||
 [14] [14] | |||
| Danger | |||
| H290, H314[14] | |||
| P280, P305+P351+P338, P310[14] | |||
| NFPA 704 (fire diamond) | |||
| Flash point | Non-flammable | ||
| Lethal dose or concentration (LD, LC): | |||
LD50 (median dose) |
1530 mg/kg (rat, oral)[15] | ||
| NIOSH (US health exposure limits): | |||
PEL (Permissible) |
TWA 1 mg/m3[10] | ||
REL (Recommended) |
TWA 1 mg/m3 ST 3 mg/m3[10] | ||
IDLH (Immediate danger) |
1000 mg/m3[10] | ||
| Safety data sheet (SDS) | ICSC 1008 | ||
| Related compounds | |||
Related phosphorus oxoacids |
|||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
Phosphoric acid (orthophosphoric acid, monophosphoric acid or phosphoric(V) acid) is a colorless, odorless phosphorus-containing solid, and inorganic compound with the chemical formula H3PO4. It is commonly encountered as an 85% aqueous solution, which is a colourless, odourless, and non-volatile syrupy liquid. It is a major industrial chemical, being a component of many fertilizers.
The compound is an acid. Removal of all three H+ ions gives the phosphate ion PO3−4. Removal of one or two protons gives dihydrogen phosphate ion H2PO−4, and the hydrogen phosphate ion HPO2−4, respectively. Phosphoric acid forms esters, called organophosphates.[16]
The name "orthophosphoric acid" can be used to distinguish this specific acid from other "phosphoric acids", such as pyrophosphoric acid. Nevertheless, the term "phosphoric acid" often means this specific compound; and that is the current IUPAC nomenclature.