
Perfluorobutanesulfonic acid
Chemical compound / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Perfluorobutanesulfonic acid?
Summarize this article for a 10 year old
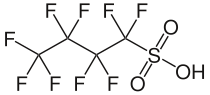
Perfluorobutanesulfonic acid (PFBS) is a PFAS chemical compound having a four-carbon fluorocarbon chain and a sulfonic acid functional group. It is stable and unreactive because of the strength of carbon–fluorine bonds. It can occur in the form of a colorless liquid or a corrosive solid.[1] Its conjugate base is perfluorobutanesulfonate (also called nonaflate) which functions as the hydrophobe in fluorosurfactants.
 | |
 | |
| Names | |
|---|---|
| Preferred IUPAC name
1,1,2,2,3,3,4,4,4-Nonafluorobutane-1-sulfonic acid | |
| Other names
FC-98 Nonaflate | |
| Identifiers | |
| |
3D model (JSmol) |
|
| ChEBI | |
| ChemSpider |
|
| ECHA InfoCard | 100.006.176 |
| EC Number |
|
PubChem CID |
|
| RTECS number |
|
| UNII | |
| UN number | 3094, 3265 |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C4HF9O3S | |
| Molar mass | 300.10 g/mol |
| Boiling point | 210–212 °C (410–414 °F; 483–485 K)[1] |
| Hazards | |
| GHS labelling: | |
  [2] [2] | |
| Danger | |
| H302, H314 | |
| P280, P305+P351+P338, P310[2] | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Since June 2003, 3M has used PFBS as a replacement for the persistent, toxic, and bioaccumulative compound perfluorooctanesulfonic acid (PFOS) in its Scotchgard stain repellents.[3] 3M markets surfactant with PFBS in two fluorosurfactants.[4]