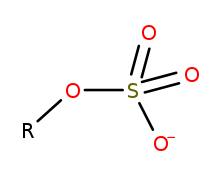
Organosulfate
Organic compounds of the form R–O–SO₃ (charge –1) / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Organosulfate?
Summarize this article for a 10 year old
In organosulfur chemistry, organosulfates are a class of organic compounds sharing a common functional group with the structure R−O−SO−3. The SO4 core is a sulfate group and the R group is any organic residue. All organosulfates are formally esters derived from alcohols and sulfuric acid (H2SO4) although many are not prepared in this way. Many sulfate esters are used in detergents, and some are useful reagents. Alkyl sulfates consist of a hydrophobic hydrocarbon chain, a polar sulfate group (containing an anion) and either a cation or amine to neutralize the sulfate group. Examples include: sodium lauryl sulfate (also known as sulfuric acid mono dodecyl ester sodium salt) and related potassium and ammonium salts.