Alcohol (chemistry)
Organic compound with at least one hydroxyl (–OH) group / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Alcohol (chemistry)?
Summarize this article for a 10 year old
SHOW ALL QUESTIONS
This article is about the class of chemical compounds. For the alcohol found in alcoholic drinks, see Alcohol (drug). For other uses, see Alcohol (disambiguation).
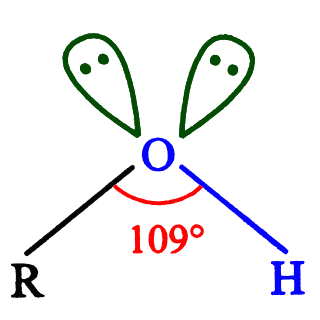
In chemistry, an alcohol is a type of organic compound that carries at least one hydroxyl (−OH) functional group bound to a saturated carbon atom.[2][3] Alcohols range from the simple, like methanol and ethanol, to complex, like sugar alcohols and cholesterol. The presence of an OH group strongly modifies the properties of hydrocarbons, conferring hydrophilic (water-loving) properties. The OH group provides a site at which many reactions can occur.