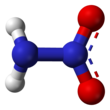
Nitramide
Chemical compound (H₂NNO₂) / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Nitramide?
Summarize this article for a 10 year old
SHOW ALL QUESTIONS
Nitramide or nitroamine is a chemical compound with the molecular formula H2N−NO2. Substituted derivatives R1R2N−NO2 are termed nitramides or nitroamines as well. Organyl derivatives of nitramide, R−NH−NO2 and R2N−NO2, are widely used as explosives: examples include RDX and HMX. It is an isomer of hyponitrous acid. Nitramide can be viewed as a nitrogen analog of nitric acid (HO−NO2), in which the hydroxyl group −OH is replaced with the amino group −NH2.
Quick Facts Names, Identifiers ...
 | |||
| |||
| Names | |||
|---|---|---|---|
| IUPAC name
Nitramide | |||
| Other names | |||
| Identifiers | |||
3D model (JSmol) |
|||
| ChEBI | |||
| ChemSpider |
| ||
PubChem CID |
|||
| UNII | |||
CompTox Dashboard (EPA) |
|||
| |||
| |||
| Properties | |||
| H2N−NO2 | |||
| Molar mass | 62.028 g·mol−1 | ||
| Appearance | Colorless solid[2] | ||
| Density | 1.378 g/cm3 | ||
| Melting point | 72 to 75 °C (162 to 167 °F; 345 to 348 K)[2] | ||
| Related compounds | |||
Related compounds |
|||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
Close