
Molybdic acid
Chemical compound / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Molybdic acid?
Summarize this article for a 10 year old
SHOW ALL QUESTIONS
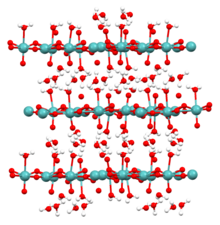
Molybdic acid refers to hydrated forms of molybdenum trioxide and related species. The monohydrate (MoO3·H2O) and the dihydrate (MoO3·2H2O) are well characterized. They are yellow diamagnetic solids.
Quick Facts Names, Identifiers ...
 | |
 | |
| Names | |
|---|---|
| Other names
Molybdic(VI) acid | |
| Identifiers | |
| |
3D model (JSmol) |
|
| ChEBI | |
| ChemSpider | |
| ECHA InfoCard | 100.029.063 |
| EC Number |
|
| KEGG | |
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| MoO3·H2O | |
| Molar mass | 161.95 g mol−1 [1] |
| Appearance | white crystals (anhydrous) yellow crystals (monohydrate) |
| Density | 3.112 g/cm3 (anhydrous) 3.124 g/cm3 (monohydrate) |
| Melting point | 300 °C (572 °F; 573 K)[1] |
| 1510 mg dm−3 Soluble in 10% ammonia 35gm/lt | |
| Structure | |
| hexagonal (anhydrous) monoclinic (monohydrate) | |
| Hazards | |
| GHS labelling:[2] | |
  | |
| Warning | |
| H319, H335, H373 | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Close