
Mercury(IV) fluoride
Chemical compound / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Mercury(IV) fluoride?
Summarize this article for a 10 year old
SHOW ALL QUESTIONS
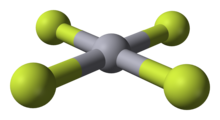
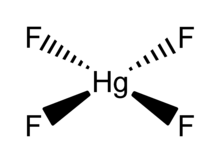
Mercury(IV) fluoride, HgF4, is the first mercury compound to be reported with mercury in the +4 oxidation state. Mercury, like the other group 12 elements (cadmium and zinc), has an s2d10 electron configuration and generally only forms bonds involving its 6s orbital. This means that the highest oxidation state mercury normally attains is +2, and for this reason it is sometimes considered a post-transition metal instead of a transition metal. HgF4 was first reported from experiments in 2007, but its existence remains disputed; experiments conducted in 2008 could not replicate the compound.[1][2]
Quick Facts Identifiers, Properties ...
 | |
 | |
| Identifiers | |
|---|---|
3D model (JSmol) |
|
| |
| |
| Properties | |
| HgF4 | |
| Molar mass | 276.58 g/mol |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Close