
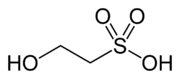
Isethionic acid
Chemical compound / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Isethionic acid?
Summarize this article for a 10 year old
SHOW ALL QUESTIONS
Isethionic acid is an organosulfur compound containing an alkylsulfonic acid located beta to a hydroxy group. Its discovery is generally attributed to Heinrich Gustav Magnus, who prepared it by the action of solid sulfur trioxide on ethanol in 1833.[1] It is a white water-soluble solid used in the manufacture of certain surfactants and in the industrial production of taurine. It is most commonly available in the form of its sodium salt (sodium isethionate).
Quick Facts Names, Identifiers ...
 | |
 Isethionic acid | |
| Names | |
|---|---|
| Preferred IUPAC name
2-Hydroxyethane-1-sulfonic acid | |
| Identifiers | |
| |
3D model (JSmol) |
|
| ChEBI | |
| ChemSpider |
|
| ECHA InfoCard | 100.003.169 |
| KEGG |
|
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C2H6O4S | |
| Molar mass | 126.13 g/mol |
| Density | 1.63 g/cm3 |
| Acidity (pKa) | 1.39 (predicted) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Close