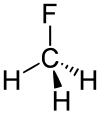
Fluoromethane
Chemical compound / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Fluoromethane?
Summarize this article for a 10 year old
SHOW ALL QUESTIONS
Fluoromethane, also known as methyl fluoride, Freon 41, Halocarbon-41 and HFC-41, is a non-toxic, liquefiable, and flammable gas at standard temperature and pressure. It is made of carbon, hydrogen, and fluorine. The name stems from the fact that it is methane (CH4) with a fluorine atom substituted for one of the hydrogen atoms. It is used in semiconductor manufacturing processes as an etching gas in plasma etch reactors.[2]
Quick Facts Names, Identifiers ...
| |||
| Names | |||
|---|---|---|---|
| IUPAC name
Fluoromethane | |||
| Other names
Freon 41 Methyl fluoride | |||
| Identifiers | |||
| |||
3D model (JSmol) |
|||
| Abbreviations | R41 | ||
| 1730725 | |||
| ChEBI | |||
| ChEMBL | |||
| ChemSpider |
| ||
| ECHA InfoCard | 100.008.907 | ||
| EC Number |
| ||
| 391 | |||
| KEGG |
| ||
| MeSH | Fluoromethane | ||
PubChem CID |
|||
| UNII | |||
| UN number | UN 2454 | ||
CompTox Dashboard (EPA) |
|||
| |||
| |||
| Properties | |||
| CH3F | |||
| Molar mass | 34.03 g/mol | ||
| Appearance | Colourless gas | ||
| Odor | pleasant, ether-like odour at high concentrations | ||
| Density | 1.4397 g/L 0.557 g/cm3 (liquid) at saturation pressure at 25 °C | ||
| Melting point | −137.8 °C (−216.0 °F; 135.3 K)[1] | ||
| Boiling point | −78.4 °C (−109.1 °F; 194.8 K)[1] | ||
| 1.66 L/kg (2.295 g/L) | |||
| Vapor pressure | 3.3 MPa | ||
| Hazards | |||
| GHS labelling: | |||
  | |||
| Danger | |||
| H220, H280 | |||
| P210, P377, P381, P403, P410+P403 | |||
| NFPA 704 (fire diamond) | |||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
Close