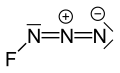Fluorine azide
Chemical compound / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Fluorine azide?
Summarize this article for a 10 year old
SHOW ALL QUESTIONS
Fluorine azide or triazadienyl fluoride is a yellow green gas composed of nitrogen and fluorine with formula FN3.[1] Its properties resemble those of ClN3, BrN3, and IN3.[2] The bond between the fluorine atom and the nitrogen is very weak, leading to this substance being very unstable and prone to explosion.[3] Calculations show the F–N–N angle to be around 102° with a straight line of 3 nitrogen atoms.[4]
Quick Facts Names, Identifiers ...
| |||
| Names | |||
|---|---|---|---|
| Other names
triazadienyl fluoride | |||
| Identifiers | |||
3D model (JSmol) |
|||
PubChem CID |
|||
CompTox Dashboard (EPA) |
|||
| |||
| |||
| Properties | |||
| FN3 | |||
| Molar mass | 61.019 g/mol | ||
| Appearance | Yellow-green gas | ||
| Melting point | −139 °C (−218 °F; 134 K) | ||
| Boiling point | −30 °C (−22 °F; 243 K) | ||
| Explosive data | |||
| Shock sensitivity | Extreme | ||
| Friction sensitivity | Extreme | ||
| Hazards | |||
| Occupational safety and health (OHS/OSH): | |||
Main hazards |
Extremely sensitive explosive | ||
| NFPA 704 (fire diamond) | |||
| Related compounds | |||
Other cations |
Hydrazoic acid Chlorine azide Bromine azide Iodine azide | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
Close
The gas boils at –30° and melts at –139 °C.[5]
It was first made by John F. Haller in 1942.[6]