
Finerenone
Chemical compound / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Finerenone?
Summarize this article for a 10 year old
Finerenone, sold under the brand name Kerendia and Firialta, is a medication used to reduce the risk of kidney function decline, kidney failure, cardiovascular death, non-fatal heart attacks, and hospitalization for heart failure in adults with chronic kidney disease associated with type 2 diabetes.[8] Finerenone is a non-steroidal mineralocorticoid receptor antagonist (MRA).[7] It is taken orally (swallowed by mouth).
 | |
| Clinical data | |
|---|---|
| Trade names | Kerendia |
| Other names | BAY 94-8862 |
| License data |
|
| Pregnancy category | |
| Routes of administration | Oral |
| Drug class | Potassium-sparing diuretic |
| ATC code | |
| Legal status | |
| Legal status | |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.247.614 |
| Chemical and physical data | |
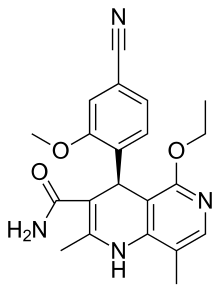
| Formula | C21H22N4O3 |
| Molar mass | 378.432 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Common side effects include hyperkalemia (high levels of potassium), hypotension (low blood pressure), and hyponatremia (low levels of sodium).[8]
Finerenone was approved for medical use in the United States in July 2021,[8][10] and in the European Union in February 2022.[9] The U.S. Food and Drug Administration considers it to be a first-in-class medication.[11]