
Ferricyanide
Anion in which a Fe3+ ion is complexed by 6 CN− ions / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Ferricyanide?
Summarize this article for a 10 year old
SHOW ALL QUESTIONS
Not to be confused with ferrocyanide.
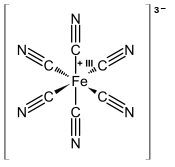
Ferricyanide is the anion [Fe(CN)6]3−. It is also called hexacyanoferrate(III) and in rare, but systematic nomenclature, hexacyanidoferrate(III). The most common salt of this anion is potassium ferricyanide, a red crystalline material that is used as an oxidant in organic chemistry.[1]
Quick Facts Names, Identifiers ...
 | |
 | |
| Names | |
|---|---|
| IUPAC name
iron(3+) hexacyanide | |
| Systematic IUPAC name
hexacyanidoferrate(III) | |
| Other names
ferric hexacyanide; hexacyanidoferrate(3−); hexacyanoferrate(III) | |
| Identifiers | |
3D model (JSmol) |
|
| ChEBI | |
| ChemSpider |
|
| KEGG | |
PubChem CID |
|
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| [Fe(CN)6]3− | |
| Related compounds | |
Other cations |
Hexacyanonickelate(III) |
Related compounds |
Ferrocyanide |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Close