
Fenthion
Chemical compound / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Fenthion?
Summarize this article for a 10 year old
SHOW ALL QUESTIONS
Fenthion is an organothiophosphate insecticide, avicide, and acaricide. Like most other organophosphates, its mode of action is via cholinesterase inhibition. Due to its relatively low toxicity towards humans and mammals, fenthion is listed as moderately toxic compound in U.S. Environmental Protection Agency and World Health Organization toxicity class.[1][3]
Quick Facts Names, Identifiers ...
 | |
| Names | |
|---|---|
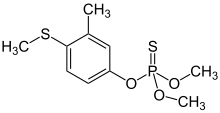
| Preferred IUPAC name
O,O-Dimethyl O-[3-methyl-4-(methylsulfanyl)phenyl] phosphorothioate | |
| Other names
• Dimethoxy-[3-methyl-4-(methylthio)phenoxy]-thioxophosphorane • O,O-Dimethyl O-[3-methyl-4-(methylthio)phenyl] phosphorothioate • O,O-Dimethyl O-4-methylthio-m-tolyl phosphorothioate | |
| Identifiers | |
| |
3D model (JSmol) |
|
| ChEBI | |
| ChEMBL | |
| ChemSpider |
|
| ECHA InfoCard | 100.000.211 |
| KEGG |
|
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C10H15O3PS2 | |
| Molar mass | 278.33 g/mol |
| Appearance | colorless, almost odorless liquid; 95-98% pure fenthion is a brown oily liquid with a weak garlic odor |
| Density | 1.250 g/cm3 (at 20 °C / 4 °C) |
| Melting point | 7 °C (45 °F; 280 K) |
| Boiling point | 87 °C (189 °F; 360 K) at 0.01 mmHg |
| 54-56 ppm (at 20 °C) | |
| Solubility in glyceride oils, methanol, ethanol, ether, acetone, and most organic solvents, especially chlorinated hydrocarbons | soluble |
| Vapor pressure | 4 • 10−5 mmHg (at 20 °C) |
| Pharmacology | |
| QP53BB02 (WHO) | |
| Hazards | |
| NIOSH (US health exposure limits): | |
PEL (Permissible) |
none[2] |
REL (Recommended) |
None established[2] |
IDLH (Immediate danger) |
N.D.[2] |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Close