
Diazomethane
Simplest diazo compound and methylating agent / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Diazomethane?
Summarize this article for a 10 year old
SHOW ALL QUESTIONS
Diazomethane is an organic chemical compound with the formula CH2N2, discovered by German chemist Hans von Pechmann in 1894. It is the simplest diazo compound. In the pure form at room temperature, it is an extremely sensitive explosive yellow gas; thus, it is almost universally used as a solution in diethyl ether. The compound is a popular methylating agent in the laboratory, but it is too hazardous to be employed on an industrial scale without special precautions.[4] Use of diazomethane has been significantly reduced by the introduction of the safer and equivalent reagent trimethylsilyldiazomethane.[5]
Quick Facts Names, Identifiers ...
 | |
 | |
| Names | |
|---|---|
| IUPAC name
Diazomethane | |
| Other names
Azimethylene, Azomethylene, | |
| Identifiers | |
| |
3D model (JSmol) |
|
| ChEBI | |
| ChemSpider |
|
| ECHA InfoCard | 100.005.803 |
| EC Number |
|
| KEGG |
|
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| CH2N2 | |
| Molar mass | 42.04 g/mol |
| Appearance | Yellow gas |
| Odor | musty |
| Density | 1.4 (air=1) |
| Melting point | −145 °C (−229 °F; 128 K) |
| Boiling point | −23 °C (−9 °F; 250 K) |
| hydrolysis[1] | |
| Structure | |
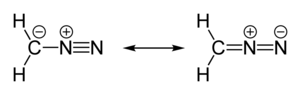
| linear C=N=N | |
| polar | |
| Hazards | |
| Occupational safety and health (OHS/OSH): | |
Main hazards |
toxic and explosive |
| GHS labelling: | |
  | |
| Danger | |
| H350 | |
| P201, P202, P281, P308+P313, P405, P501 | |
| NFPA 704 (fire diamond) | |
| Lethal dose or concentration (LD, LC): | |
LC50 (median concentration) |
175 ppm (cat, 10 min)[2] |
| NIOSH (US health exposure limits): | |
PEL (Permissible) |
TWA 0.2 ppm (0.4 mg/m3)[3] |
REL (Recommended) |
TWA 0.2 ppm (0.4 mg/m3)[3] |
IDLH (Immediate danger) |
2 ppm[3] |
| Related compounds | |
Related functional groups; compounds |
R-N=N=N (azide), R-N=N-R (azo); R2CN2 R = Ph, tms, CF3 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Close
