
Dapagliflozin
Diabetes medication / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Dapagliflozin?
Summarize this article for a 10 year old
Dapagliflozin, sold under the brand names Farxiga (US) and Forxiga (EU) among others, is a medication used to treat type 2 diabetes.[6][7][9] It is also used to treat adults with heart failure and chronic kidney disease.[10][11][7] It reversibly inhibits sodium-glucose co-transporter 2 (SGLT-2) in the renal proximal convoluted tubule to reduce glucose reabsorption and increase urinary glucose excretion.[12]
 | |||
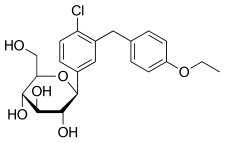 Haworth projection (bottom) | |||
| |||
| Clinical data | |||
|---|---|---|---|
| Pronunciation | /ˌdæpəɡlɪˈfloʊzɪn/ DAP-ə-glif-LOH-zin | ||
| Trade names | Forxiga, Farxiga, others | ||
| Other names | BMS-512148; (1S)-1,5-anhydro-1-C-{4-chloro-3-[(4-ethoxyphenyl)methyl]phenyl}-D-glucitol | ||
| AHFS/Drugs.com | Monograph | ||
| MedlinePlus | a614015 | ||
| License data | |||
| Pregnancy category |
| ||
| Routes of administration | By mouth | ||
| Drug class | Sodium-glucose co-transporter 2 (SGLT2) inhibitor | ||
| ATC code | |||
| Legal status | |||
| Legal status | |||
| Pharmacokinetic data | |||
| Bioavailability | 78% (after 10 mg dose) | ||
| Protein binding | ~91% | ||
| Metabolism | UGT1A9 (major), CYP (minor) | ||
| Metabolites | Dapagliflozin 3-O-glucuronide (inactive) | ||
| Elimination half-life | ~12.9 hours | ||
| Excretion | Urine (75%), feces (21%)[6] | ||
| Identifiers | |||
| |||
| CAS Number | |||
| PubChem CID | |||
| IUPHAR/BPS | |||
| DrugBank | |||
| ChemSpider | |||
| UNII | |||
| KEGG | |||
| ChEBI | |||
| ChEMBL | |||
| CompTox Dashboard (EPA) | |||
| ECHA InfoCard | 100.167.331 | ||
| Chemical and physical data | |||
| Formula | C21H25ClO6 | ||
| Molar mass | 408.88 g·mol−1 | ||
| 3D model (JSmol) | |||
| |||
| |||
Common side effects include hypoglycaemia (low blood sugar), urinary tract infections, genital infections, and volume depletion (reduced amount of water in the body).[13] Diabetic ketoacidosis is a common side effect in people with type 1 diabetes.[14] Serious but rare side effects include Fournier gangrene.[15]
It was developed by Bristol-Myers Squibb in partnership with AstraZeneca. It is on the World Health Organization's List of Essential Medicines.[16] In 2021, it was the 187th most commonly prescribed medication in the United States, with more than 2 million prescriptions.[17][18] Dapagliflozin is available as a generic medication.
