
Choline
Chemical compound and essential nutrient / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Choline?
Summarize this article for a 10 year old
SHOW ALL QUESTIONS
Not to be confused with chlorine.
Choline (/ˈkoʊliːn/ KOH-leen)[4] is an essential nutrient for humans and many other animals, which was formerly classified as a B vitamin (vitamin B4).[5][6] It is a structural part of phospholipids and a methyl donor in metabolic one-carbon chemistry. The compound is related to trimethylglycine in the latter respect. It is a cation with the chemical formula [(CH3)3NCH2CH2OH]+. Choline forms various salts, for example choline chloride and choline bitartrate.
Quick Facts Names, Identifiers ...
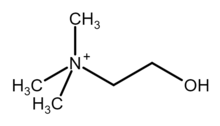 | |
 | |
| Names | |
|---|---|
| IUPAC name
2-Hydroxyethyl(trimethyl)azanium[1] | |
| Preferred IUPAC name
2-Hydroxy-N,N,N-trimethylethan-1-aminium | |
Other names
| |
| Identifiers | |
| |
3D model (JSmol) |
|
| 1736748 | |
| ChEBI | |
| ChEMBL | |
| ChemSpider |
|
| DrugBank |
|
| ECHA InfoCard | 100.000.487 |
| EC Number |
|
| 324597 | |
| KEGG |
|
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| [(CH3)3NCH2CH2OH]+ | |
| Molar mass | 104.173 g·mol−1 |
| Appearance | Viscous colorless deliquescent liquid (choline hydroxide)[2] |
| Very soluble (choline hydroxide)[2] | |
| Solubility | soluble in ethanol,[2] insoluble in diethylether and chloroform[3] (choline hydroxide) |
| Structure | |
| Tetrahedral at the nitrogen atom | |
| Hazards | |
| Occupational safety and health (OHS/OSH): | |
Main hazards |
Corrosive |
| GHS labelling: | |
 | |
| Danger | |
| H314 | |
| P260, P264, P280, P301+P330+P331, P303+P361+P353, P304+P340, P305+P351+P338, P310, P321, P363, P405, P501 | |
| NFPA 704 (fire diamond) | |
| Lethal dose or concentration (LD, LC): | |
LD50 (median dose) |
3–6 g/kg (rat, oral)[2] |
| Safety data sheet (SDS) | 4 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Close
