
Chloroplatinic acid
Chemical compound / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Chloroplatinic acid?
Summarize this article for a 10 year old
SHOW ALL QUESTIONS
Chloroplatinic acid (also known as hexachloroplatinic acid) is an inorganic compound with the formula [H3O]2[PtCl6](H2O)x (0 ≤ x ≤ 6). A red solid, it is an important commercial source of platinum, usually as an aqueous solution. Although often written in shorthand as H2PtCl6, it is the hydronium (H3O+) salt of the hexachloroplatinate anion (PtCl2−
6).[1][2][3] Hexachloroplatinic acid is highly hygroscopic.
Quick Facts Names, Identifiers ...
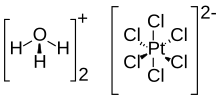 | |
 | |
 | |
| Names | |
|---|---|
| IUPAC name
Dihydronium hexachloroplatinate(2–) | |
| Other names
Hexachloroplatinic acid Hydronium hexachloroplatinate(IV) | |
| Identifiers | |
3D model (JSmol) |
|
| ChemSpider |
|
| ECHA InfoCard | 100.037.267 |
| EC Number |
|
PubChem CID |
|
| RTECS number |
|
| UNII | |
| UN number | 2507 |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| H6Cl6Pt | |
| Molar mass | 409.81 g/mol |
| Appearance | Reddish brown solid |
| Density | 2.431 g/cm3 |
| Melting point | 60 °C (140 °F; 333 K) |
| Boiling point | decomposes |
| highly soluble | |
| Structure | |
| Anti-fluorite. | |
| octahedral | |
| 0 D | |
| Hazards | |
| GHS labelling: | |
    | |
| Danger | |
| H301, H314, H317, H334 | |
| P260, P261, P264, P270, P272, P280, P285, P301+P310, P301+P330+P331, P302+P352, P303+P361+P353, P304+P340, P304+P341, P305+P351+P338, P310, P321, P330, P333+P313, P342+P311, P363, P405, P501 | |
| NFPA 704 (fire diamond) | |
| Safety data sheet (SDS) | External MSDS |
| Related compounds | |
Other anions |
Hexachloropalladic acid |
Other cations |
Potassium hexachloroplatinate, Ammonium hexachloroplatinate, Rubidium hexachloroplatinate, Caesium hexachloroplatinate |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Close
