
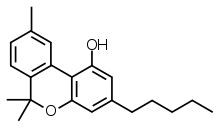
Cannabinol
Naturally-occurring cannabinoid / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Cannabinol?
Summarize this article for a 10 year old
Cannabinol (CBN) is a mildly psychoactive cannabinoid (e.g., CBD) that acts as a low affinity partial agonist at both CB1 and CB2 receptors. This activity at CB1 and CB2 receptors constitutes interaction of CBN with the endocannabinoid system (ECS).
 | |
 | |
| Legal status | |
|---|---|
| Legal status |
|
| Identifiers | |
| |
| CAS Number |
|
| PubChem CID | |
| IUPHAR/BPS | |
| ChemSpider |
|
| UNII | |
| KEGG |
|
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.216.772 |
| Chemical and physical data | |
| Formula | C21H26O2 |
| Molar mass | 310.437 g·mol−1 |
| 3D model (JSmol) | |
| Melting point | 76–77 °C (169–171 °F) [1] |
| Solubility in water | Insoluble in water,[2] soluble in methanol[3] and ethanol[4] mg/mL (20 °C) |
| |
| |
| | |
In 1896 Cannabinol was first discovered in Cannabis by Thomas Barlow Wood, W.T Newton Spivey, and Thomas Easterfield.[5] In the early 1930s CBNs structure was identified by Robert Sidney Cahn,[6][7] marking the first development of a cannabis extract.
Its chemical synthesis were achieved by 1940, followed by some of the first basic research studies to determine the effects of individual cannabis-derived compounds in vivo. Although CBN shares the same mechanism of action as other phytocannabinoids (e.g., Delta-9-tetrahydrocannabinol, Δ9-THC), it has a lower affinity for CB1 receptors, meaning that much higher doses of CBN are required in order to experience effects, such as mild sedation.