COVIran Barekat
COVIran Barakat vaccine against COVID-19 / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about COVIran Barekat?
Summarize this article for a 10 year old
COVIran Barekat[2] (Persian: کووایران برکت) is a COVID-19 vaccine developed in Iran by Shifa Pharmed Industrial Group, a subsidiary of the Barkat Pharmaceutical Group.[3][4][5] It is an inactivated virus-based vaccine.[2] Iranian authorities have authorized its emergency use. This makes it the first locally developed COVID-19 vaccine to be approved for emergency use in the Middle East.[6]
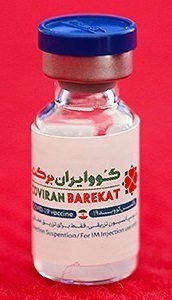 A vial of COVIran Barakat | |
| Vaccine description | |
|---|---|
| Target | SARS-CoV-2 |
| Vaccine type | Inactivated |
| Clinical data | |
| Other names | BIV1-CovIran[1] COVIran Barakat COVIran Barkat کووایران برکت |
| Routes of administration | Intramuscular |
| ATC code |
|
| Legal status | |
| Legal status |
Full and emergency authorizations Full list of COVIran Barekat authorizations |
Officials in charge say they are in the process to publish the results of the clinical trials in a peer-reviewed journal.[7] The interim results of the phases 1 and 2 trials showed 93.5% (95% CI, 88.4–99.6%) of the receivers of the vaccine have produced neutralizing antibodies against SARS-CoV-2.[8] Those results have not been peer-reviewed and describe the immunogenicity of the vaccine and not its efficacy. On 3 March 2022, peer-reviewed results have been published in the Clinical Microbiology and Infection.[9] [further explanation needed]
As of February 27, 2022, approximately 60 million doses have been produced according to Shifa Pharmed's CEO.[10] and the annual production capacity of this vaccine has reached 350 million doses.[11] Multiple Iranian personalities have received the vaccine, including the Supreme Leader, Ali Khamenei[4] and the President, Ebrahim Raisi.[12]
A version of the vaccine based on the Omicron variant and named CovIran Barkat Plus is in development and studied to be given as a third dose.[13][14]