
Bicalutamide
Prostate cancer treatment / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Bicalutamide?
Summarize this article for a 10 year old
Bicalutamide, sold under the brand name Casodex among others, is an antiandrogen medication that is primarily used to treat prostate cancer.[10] It is typically used together with a gonadotropin-releasing hormone (GnRH) analogue or surgical removal of the testicles to treat metastatic prostate cancer (mPC).[11][10][12] To a lesser extent, it is used at high doses for locally advanced prostate cancer (LAPC) as a monotherapy without castration.[4][2][13] Bicalutamide was also previously used as monotherapy to treat localized prostate cancer (LPC), but authorization for this use was withdrawn following unfavorable trial findings.[13][14][15][16] Besides prostate cancer, bicalutamide is limitedly used in the treatment of excessive hair growth and scalp hair loss in women,[17][18] as a puberty blocker and component of feminizing hormone therapy for transgender girls and women,[19] to treat gonadotropin-independent early puberty in boys,[20] and to prevent overly long-lasting erections in men.[21] It is taken by mouth.[10]
 | |
 | |
| Clinical data | |
|---|---|
| Pronunciation | Bicalutamide: • /ˌbaɪkəˈluːtəmaɪd/[1] • BY-kə-LOO-tə-myde[1] |
| Trade names | Casodex, Calutex, others |
| Other names | ICI-176,334; ZD-176,334 |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a697047 |
| License data |
|
| Pregnancy category |
|
| Routes of administration | By mouth[2] |
| Drug class | Nonsteroidal antiandrogen |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | Well-absorbed; absolute bioavailability unknown[3] |
| Protein binding | Racemate: 96.1%[2] (R)-Isomer: 99.6%[2] (Mainly to albumin)[2] |
| Metabolism | Liver (extensively):[4][5] • Hydroxylation (CYP3A4) • Glucuronidation (UGT1A9) |
| Metabolites | • Bicalutamide glucuronide • Hydroxybicalutamide • Hydroxybicalutamide gluc. (All inactive)[4][2][6][7] |
| Elimination half-life | Single-dose: 5.8 days[8] Continuous: 7–10 days[9] |
| Excretion | Feces: 43%[4] Urine: 34%[4] |
| Identifiers | |
| |
| CAS Number |
|
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank |
|
| ChemSpider |
|
| UNII | |
| KEGG |
|
| ChEBI | |
| ChEMBL | |
| PDB ligand | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.126.100 |
| Chemical and physical data | |
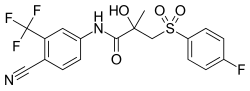
| Formula | C18H14F4N2O4S |
| Molar mass | 430.37 g·mol−1 |
| 3D model (JSmol) | |
| Chirality | Racemic mixture (of (R)- and (S)-enantiomers) |
| Melting point | 191 to 193 °C (376 to 379 °F) (experimental) |
| Boiling point | 650 °C (1,202 °F) (predicted) |
| Solubility in water | 0.005 |
| |
| |
| (verify) | |
Common side effects of bicalutamide in men include breast growth, breast tenderness, and hot flashes.[10] Other side effects in men include feminization and sexual dysfunction.[22][23] Some side effects like breast changes and feminization are minimal when combined with castration.[24] While the medication appears to produce few side effects in women, its use in women is not explicitly approved by the Food and Drug Administration (FDA) at this time.[25][10] Use during pregnancy may harm the baby.[10] In men with early prostate cancer, bicalutamide monotherapy has been found to increase the likelihood of death from causes other than prostate cancer.[26][13] Bicalutamide produces abnormal liver changes necessitating discontinuation in around 1% of people.[27][13] Rarely, it has been associated with cases of serious liver damage,[10] serious lung toxicity,[3] and sensitivity to light.[28][29] Although the risk of adverse liver changes is small, monitoring of liver function is recommended during treatment.[10]
Bicalutamide is a member of the nonsteroidal antiandrogen (NSAA) group of medications.[3] It works by selectively blocking the androgen receptor (AR), the biological target of the androgen sex hormones testosterone and dihydrotestosterone (DHT).[30] It does not lower androgen levels.[3] The medication can have some estrogen-like effects in men when used as a monotherapy due to increased estradiol levels.[31][32][33] Bicalutamide is well-absorbed, and its absorption is not affected by food.[2] The elimination half-life of the medication is around one week.[2][10] It shows peripheral selectivity in animals, but crosses the blood–brain barrier and affects both the body and brain in humans.[2][34]
Bicalutamide was patented in 1982 and approved for medical use in 1995.[35] It is on the World Health Organization's List of Essential Medicines.[36] Bicalutamide is available as a generic medication.[37] The drug is sold in more than 80 countries, including most developed countries.[38][39][40] It was at one time the most widely used antiandrogen in the treatment of prostate cancer, with millions of men with the disease having been prescribed it.[23][41][42][43][44] Although bicalutamide is also used for other indications besides prostate cancer, the vast majority of prescriptions appear to be for treatment of prostate cancer.[44]