
Azelaic acid
Organic chemical compound / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Azelaic acid?
Summarize this article for a 10 year old
SHOW ALL QUESTIONS
"Finevin" redirects here. The term may also refer to a former settlement in Sutherland, Scotland.
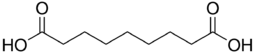
Azelaic acid (AzA) is an organic compound with the formula HOOC(CH2)7COOH.[3] This saturated dicarboxylic acid exists as a white powder. It is found in wheat, rye, and barley. It is a precursor to diverse industrial products including polymers and plasticizers, as well as being a component of a number of hair and skin conditioners.[4] AzA inhibits tyrosinase.[5]
Quick Facts Names, Identifiers ...
 | |
 | |
| Names | |
|---|---|
| Preferred IUPAC name
Nonanedioic acid | |
| Identifiers | |
| |
3D model (JSmol) |
|
| 1101094 | |
| ChEBI | |
| ChEMBL | |
| ChemSpider |
|
| DrugBank |
|
| ECHA InfoCard | 100.004.246 |
| EC Number |
|
| 261342 | |
| KEGG |
|
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C9H16O4 | |
| Molar mass | 188.22 g/mol |
| Appearance | white solid |
| Density | 1.443 g/mL |
| Melting point | 109 to 111 °C (228 to 232 °F; 382 to 384 K)[1] |
| Boiling point | 286 °C (547 °F; 559 K) at 100 mmHg[1] |
| 2.14 g/L[2] | |
| Acidity (pKa) | 4.550, 5.498[2] |
| Pharmacology | |
| D10AX03 (WHO) | |
| Topical | |
| Pharmacokinetics: | |
| Very low | |
| 12 h | |
| Legal status | |
| Hazards | |
| GHS labelling: | |
 | |
| Warning | |
| H315, H319 | |
| P264, P280, P302+P352, P305+P351+P338, P321, P332+P313, P337+P313, P362 | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Close