
Tetraethylammonium
Polyatomic ion (N(C₂H₅)₄, charge +1) / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Tetraethylammonium?
Summarize this article for a 10 year old
SHOW ALL QUESTIONS
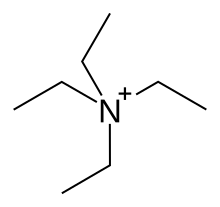
Tetraethylammonium (TEA) is a quaternary ammonium cation with the chemical formula [Et4N]+, consisting of four ethyl groups (−C2H5, denoted Et) attached to a central nitrogen atom. It is a counterion used in the research laboratory to prepare lipophilic salts of inorganic anions. It is used similarly to tetrabutylammonium, the difference being that its salts are less lipophilic, more easily crystallized and more toxic.
Quick Facts Names, Identifiers ...
 | |
 | |
| Names | |
|---|---|
| Preferred IUPAC name
N,N,N-Triethylethanaminium | |
| Other names
Tetraethylazanium | |
| Identifiers | |
| |
3D model (JSmol) |
|
| ChEBI | |
| ChEMBL | |
| ChemSpider |
|
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C8H20N+ | |
| Molar mass | 130.25 g/mol |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Close