Phosphite ester
Organic compound with the formula P(OR)3 / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Phosphite?
Summarize this article for a 10 year old
SHOW ALL QUESTIONS
"Phosphite" redirects here. For the anion, see Phosphite anion.
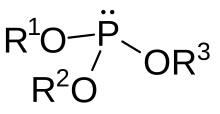
In organic chemistry, a phosphite ester, phosphorite(III) or organophosphite usually refers to an organophosphorous compound with the formula P(OR)3. They can be considered as esters of an unobserved tautomer phosphorous acid, H3PO3, with the simplest example being trimethylphosphite, P(OCH3)3. Some phosphites can be considered esters of the dominant tautomer of phosphorous acid (HP(O)(OH)2). The simplest representative is dimethylphosphite with the formula HP(O)(OCH3)2. Both classes of phosphites are usually colorless liquids.