
Olanzapine
Atypical antipsychotic medication / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Olanzapine pamoate?
Summarize this article for a 10 year old
Olanzapine, sold under the brand name Zyprexa among others, is an atypical antipsychotic primarily used to treat schizophrenia and bipolar disorder.[10] For schizophrenia, it can be used for both new-onset disease and long-term maintenance.[10] It is taken by mouth or by injection into a muscle.[10]
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Zyprexa, Zypine, others[1] |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a601213 |
| License data |
|
| Pregnancy category |
|
| Routes of administration | By mouth, intramuscular injection |
| Drug class | Atypical antipsychotic |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | 60–65%[5][6][7] |
| Protein binding | 93%[8] |
| Metabolism | Liver (direct glucuronidation and CYP1A2 mediated oxidation) |
| Elimination half-life | 33 hours, 51.8 hours (elderly)[8] |
| Excretion | Urine (57%; 7% as unchanged drug), faeces (30%)[8][9] |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank |
|
| ChemSpider |
|
| UNII | |
| KEGG |
|
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.125.320 |
| Chemical and physical data | |
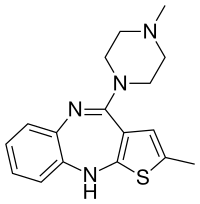
| Formula | C17H20N4S |
| Molar mass | 312.44 g·mol−1 |
| 3D model (JSmol) | |
| Melting point | 195 °C (383 °F) |
| Solubility in water | Practically insoluble in water mg/mL (20 °C) |
| |
| |
| | |
Common side effects include weight gain, movement disorders, dizziness, feeling tired, constipation, and dry mouth.[10] Other side effects include low blood pressure with standing, allergic reactions, neuroleptic malignant syndrome, high blood sugar, seizures, and tardive dyskinesia.[10] In older people with dementia, its use increases the risk of death.[10] Use in the later part of pregnancy may result in a movement disorder in the baby for some time after birth.[10] Although how it works is not entirely clear, it blocks dopamine and serotonin receptors.[10]
Olanzapine was patented in 1991 and approved for medical use in the United States in 1996.[10][11] It is available as a generic medication.[10] In 2021, it was the 164th most commonly prescribed medication in the United States, with more than 3 million prescriptions.[12][13] Eli Lilly also markets olanzapine in a fixed-dose combination with fluoxetine as olanzapine/fluoxetine (Symbyax).[14] It is on the World Health Organization's List of Essential Medicines.[15]