Nitrite
Portmanteau name for nitrite derivatives / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Nitrites?
Summarize this article for a 10 year old
SHOW ALL QUESTIONS
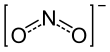
The nitrite ion has the chemical formula NO−
2. Nitrite (mostly sodium nitrite) is widely used throughout chemical and pharmaceutical industries.[1] The nitrite anion is a pervasive intermediate in the nitrogen cycle in nature. The name nitrite also refers to organic compounds having the –ONO group, which are esters of nitrous acid.
Quick Facts Names, Identifiers ...
| |||
| Names | |||
|---|---|---|---|
| IUPAC name
Nitrite | |||
| Systematic IUPAC name
dioxidonitrate(1−) | |||
| Other names
nitrite | |||
| Identifiers | |||
3D model (JSmol) |
|||
| ChEBI | |||
| ChemSpider | |||
| EC Number |
| ||
PubChem CID |
|||
| UNII | |||
| |||
| |||
| Properties | |||
| NO− 2 | |||
| Molar mass | 46.005 g·mol−1 | ||
| Conjugate acid | Nitrous acid | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
Close