Methanediol
Organic compound (CH2(OH)2); simplest geminal diol / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Methylene glycol?
Summarize this article for a 10 year old
SHOW ALL QUESTIONS
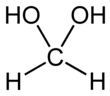
Methanediol, also known as formaldehyde monohydrate or methylene glycol, is an organic compound with chemical formula CH2(OH)2. It is the simplest geminal diol. In aqueous solutions it coexists with oligomers (short polymers). The compound is closely related and convertible to the industrially significant derivatives paraformaldehyde ((CH2O)n), formaldehyde (H2C=O), and 1,3,5-trioxane ((CH2O)3).[3]
Quick Facts Names, Identifiers ...
| |||
 | |||
| Names | |||
|---|---|---|---|
| Preferred IUPAC name
Methanediol[1] | |||
Other names
| |||
| Identifiers | |||
| |||
3D model (JSmol) |
|||
| Abbreviations | MADOL | ||
| 1730798 | |||
| ChEBI | |||
| ChemSpider |
| ||
| ECHA InfoCard | 100.006.673 | ||
| EC Number |
| ||
PubChem CID |
|||
| UNII | |||
CompTox Dashboard (EPA) |
|||
| |||
| |||
| Properties | |||
| CH4O2 | |||
| Molar mass | 48.041 g·mol−1 | ||
| Appearance | Colourless liquid | ||
| Density | 1.199 g/cm3 [citation needed] | ||
| Boiling point | 194 °C (381 °F; 467 K) at 101 kPa [citation needed] | ||
| Vapor pressure | 16.1 Pa [citation needed] | ||
| Acidity (pKa) | 13.29[2] | ||
Refractive index (nD) |
1.401 [citation needed] | ||
| Hazards | |||
| Flash point | 99.753 °C (211.555 °F; 372.903 K) | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
Close
Methanediol is a product of the hydration of formaldehyde. The equilibrium constant for hydration is estimated to be 103,[4]CH2(OH)2 predominates in dilute (<0.1%) solution. In more concentrated solutions, it oligomerizes to HO(CH2O)nH.[3]