
Trioxidane
Chemical compound / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Hydrogen trioxide?
Summarize this article for a 10 year old
SHOW ALL QUESTIONS
"Hydrogen trioxide" redirects here. For HO3, see hydrogen ozonide.
Not to be confused with Trioxane.
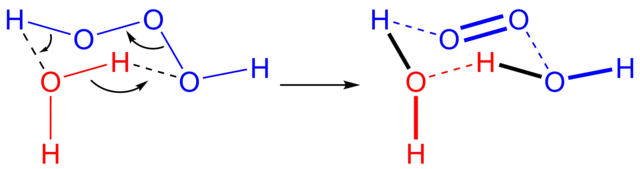
Trioxidane (systematically named dihydrogen trioxide,[2][3]), also called hydrogen trioxide[4][5] is an inorganic compound with the chemical formula H[O]
3H (can be written as [H(μ-O
3)H] or [H
2O
3]). It is one of the unstable hydrogen polyoxides.[4] In aqueous solutions, trioxidane decomposes to form water and singlet oxygen:
Quick Facts Names, Identifiers ...
 | |
 | |
| Names | |
|---|---|
| Preferred IUPAC name
Trioxidane (only preselected name)[1] | |
| Systematic IUPAC name
Dihydrogen trioxide | |
| Other names
Hydrogen trioxide Dihydroxy ether | |
| Identifiers | |
3D model (JSmol) |
|
| ChEBI | |
| ChemSpider |
|
| 200290 | |
PubChem CID |
|
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| H2O3 | |
| Molar mass | 50.013 g·mol−1 |
| Related compounds | |
Related compounds |
Hydrogen peroxide; Hydrogen ozonide; Hydroperoxyl |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Close
The reverse reaction, the addition of singlet oxygen to water, typically does not occur in part due to the scarcity of singlet oxygen. In biological systems, however, ozone is known to be generated from singlet oxygen, and the presumed mechanism is an antibody-catalyzed production of trioxidane from singlet oxygen.[2]