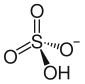
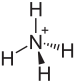Ammonium bisulfate
Chemical compound / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Ammonium bisulfate?
Summarize this article for a 10 year old
SHOW ALL QUESTIONS
Ammonium bisulfate, also known as ammonium hydrogen sulfate, is a white, crystalline solid with the formula (NH4)HSO4. This salt is the product of the half-neutralization of sulfuric acid by ammonia.
Quick Facts Names, Identifiers ...
| |||
 | |||
 | |||
| Names | |||
|---|---|---|---|
| IUPAC name
Ammonium hydrogen sulfate | |||
| Identifiers | |||
3D model (JSmol) |
|||
| ChemSpider |
| ||
| ECHA InfoCard | 100.029.332 | ||
PubChem CID |
|||
| RTECS number |
| ||
| UNII | |||
CompTox Dashboard (EPA) |
|||
| |||
| |||
| Properties | |||
| (NH4)HSO4 | |||
| Molar mass | 115.11 g/mol | ||
| Appearance | White solid | ||
| Density | 1.78 g/cm3 | ||
| Melting point | 147 °C (297 °F; 420 K) | ||
| Very soluble | |||
| Solubility in other solvents | Soluble in methanol insoluble in acetone | ||
| Hazards | |||
| NFPA 704 (fire diamond) | |||
| Safety data sheet (SDS) | External MSDS | ||
| Related compounds | |||
Other anions |
Ammonium thiosulfate Ammonium sulfite Ammonium sulfate Ammonium persulfate | ||
Other cations |
Sodium bisulfate Potassium bisulfate | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
Close