
2,6-Diaminopurine
Chemical compound / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about 2,6-diaminopurine?
Summarize this article for a 10 year old
SHOW ALL QUESTIONS
2,6-diaminopurine (2,6-DAP, also known as 2-aminoadenine) is a compound once used in the treatment of leukemia.[1] As the Z base, it is found instead of adenine (A) in the genetic material of some bacteriophage viruses.[2]
Quick Facts Names, Identifiers ...
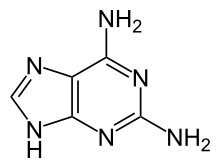 | |
| Names | |
|---|---|
| IUPAC name
7H-purine-2,6-diamine | |
| Other names
2-aminoadenine; 2,6-DAP | |
| Identifiers | |
3D model (JSmol) |
|
| ChEMBL |
|
| ChemSpider |
|
| ECHA InfoCard | 100.016.006 |
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C5H6N6 | |
| Molar mass | 150.145 g·mol−1 |
| Appearance | White to yellow crystalline powder |
| Density | 1.743 g/cm3 |
| Melting point | 117 to 122 °C (243 to 252 °F; 390 to 395 K) |
| 2.38 g/L at 20 °C | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Close
In August 2011, a report, based on NASA studies with meteorites found on Earth, was published suggesting 2,6-diaminopurine and related organic molecules, including the DNA and RNA components adenine and guanine, may have been formed extraterrestrially in outer space.[3][4][5]